|
5/7/2023 0 Comments Co2 molecular geometry 
In contrast, the Oxygen atom hybridizes to form three sp2 hybrid orbitals. Here the 2s orbitals and one of the p-orbitals will hybridize to form 2 sp orbitals. In its excited state, the atom’s electronic configuration becomes 1s2 2s1 2p3, so now every p-orbital of the atoms has one electron each. Total number of valence electrons in the molecule = 16 Valence electrons in Oxygen: 6*2 = 12 ( as there are two Oxygen atoms in the molecule, we will multiply it by 2).To know the bond formation and the arrangement, let’s go through the valence electrons of all the atoms in the molecule. Two Oxygen atoms are located on the terminals where both these atoms share electrons and form bonds with the central Carbon atom. In CO2, the Carbon atom is in the central position as it is the least electronegative atom in the molecule. Now that you know how the Lewis structure is drawn and its uses let us quickly look at the CO2 Lewis structure. Such structure helps in understanding the arrangement of atoms along with the electrons participating in the bond formation. 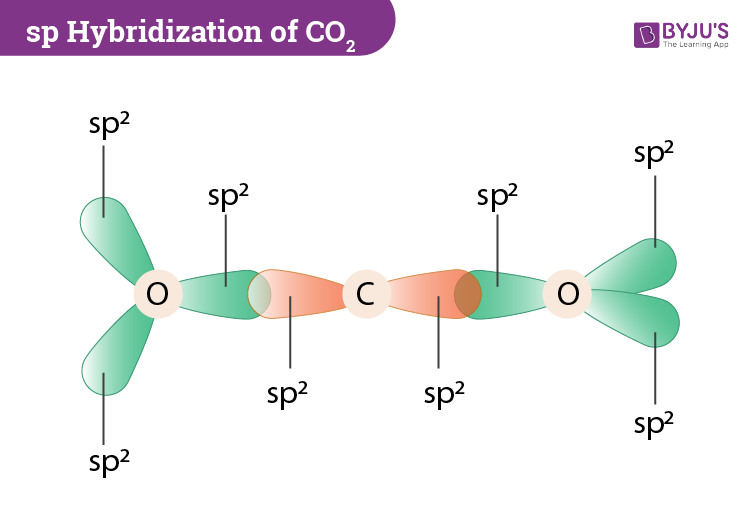
Drawing lines represent the bonds formed in the molecule. These valence electrons are represented by drawing dots around the individual atoms, hence the Lewis dot structure. Lewis dot structure is a pictorial representation of the arrangement of the valence shell electrons in the molecule. To know the lewis structure of CO2, one should first understand what precisely the Lewis structure is. This structure helps in knowing the arrangement of electrons in the molecules and the shape of the molecule. A.One needs to know the Lewis structure in order to understand the molecular geometry of any given molecule. B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. Medvedev, V.A.CODATA Key Values for Thermodynamics. 223 (1997) 59-98Ĭ Graham, DA Imrie, RE Raab "Measurement of the electric quadrupole moments of CO2, CO, N2, Cl2 and BF3" Mol. TN Olney, NM Cann, G Cooper, CE Brion, Absolute scale determination for photoabsorption spectra and the calculation of molecular properties using dipole sum-rules, Chem. "Vibrational Intensities in Infrared and Raman Spectroscopy" WB Person, G Zerbi, ed. Bond Moment constants in CO2, OCS, CS2, CSe2, and SCSe" J. PR Davies, WJ Orville-Thomas "Infrared Band Intensities and Bond polaritites Part1. Herzberg, G., Electronic spectra and electronic structure of polyatomic molecules,Van Nostrand,New York, 1966 Infrared and Raman Spectra of polyatomic molecules" 1945 Van Nostrand. 
Please address comments about this page to Herzberg" Molecular Spectra and Molecular Structure II. NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites.įurther, NIST does not endorse any commercial products that may be mentioned on these sites. There may be other web sites that are more appropriate for your purpose. No inferences should be drawn on account of other sites being referenced, or not, from this page. We have provided these links to other web sites because they may have information that would be of interest to you. You are here: Experimental > One molecule all propertiesĮxperimental data for CO 2 (Carbon dioxide)Īnhydride carbonique Carbon dioxide Carbon oxide (CO2) Carbonic acid, gas Carbonic anhydride Carbonice Dry ice Kohlendioxyd Kohlensaure īy selecting the following links, you may be leaving NIST webspace. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
